
How do you know a potential China factory can actually deliver what your customers expect, before you place a large order and send a deposit? That question is the real starting point for a successful China factory audit. A factory audit is not a paperwork exercise. It is an early risk-control step that helps importers verify whether a supplier can meet quality, capacity, compliance, and communication requirements in real operating conditions.
For U.S. brands and sourcing teams, this matters because mistakes made before production are usually the most expensive to fix later. A structured factory audit can uncover gaps in process discipline, staffing, quality controls, or certifications before they become delayed shipments, chargebacks, or rejected goods. It also supports better internal decisions across procurement, finance, and operations.
This article focuses on how to conduct that audit effectively, not just how to schedule one. In practical terms, your approach should combine three goals from day one:
Verify operational reality, not promises Confirm that the factory floor, equipment, QC checkpoints, and management systems match what was discussed during sourcing.
Align audit scope with product risk Product category drives scrutiny. For example, products with U.S. regulatory exposure, including some health and wellness items, require tighter checks against documentation and process controls linked to FDA expectations.
Use findings to make a sourcing decision Audit outputs should help you decide whether to approve, conditionally approve, or reject a supplier, and define corrective actions with deadlines.
Many buyers begin with online factory audit reviews and supplier profiles, then assume that desktop research is enough. It is useful, but incomplete. The purpose of on-site or professionally managed factory audit service support is to validate what cannot be confirmed through photos, chat messages, or sales presentations alone. This is where a reliable China audits and evaluation partner can reduce blind spots during factory audit sourcing.
Cost is another common concern. Factory audit cost and factory audit pricing vary by location, audit depth, product complexity, and reporting detail. Instead of choosing the cheapest option, treat pricing as part of your total risk budget. A narrowly scoped audit may save money upfront but miss critical issues that create much larger downstream losses.
As we move through this guide, you will learn how to define audit objectives, select the right checklist, interpret findings, and turn results into clear supplier decisions. If you are evaluating support from a China Factory Audits and evaluation service provider, keep the same standard: the process should give you decision-quality evidence, not generic pass/fail language.

After introducing what a factory audit is, the practical question is why it matters so much before you place production orders. For US importers, a China factory audit is the control point that turns sourcing from guesswork into a managed business process.
China sourcing can fail in quiet ways long before a shipment is late. A supplier may subcontract without approval, run outdated equipment, or lack consistent incoming material checks. A structured factory audit helps you verify real operating conditions, not just sales promises.
In practice, risk review should cover:
Capacity realism: Confirm whether staffing, lines, and shifts can meet your delivery windows.
Quality system maturity: Check inspection records, defect handling, and corrective action discipline.
Process stability: Verify whether key steps are standardized and documented across shifts.
Sub-supplier control: Identify who provides critical materials and how those partners are managed.
Business continuity: Assess backup plans for power, labor shortages, and tooling failures.
When teams compare factory audit reviews across candidate suppliers, they make better sourcing decisions and reduce costly surprises later.
For consumer-facing US brands, one quality failure can trigger chargebacks, returns, and marketplace penalties. A factory audit service reduces that exposure by checking whether production controls support consistent product performance.
Brand protection is not only about defects. It also includes packaging accuracy, labeling discipline, and traceability, especially for products sold on major platforms. If a factory cannot produce reliable records, your brand bears the reputational risk when complaints appear online.
This is where factory audit sourcing decisions become strategic. Selecting a partner based on verified operational capability, not only sample quality, lowers the chance that one weak shipment damages years of brand building.
Compliance gaps can stop products at the border or create legal risk after launch. During a China factory audit, buyers should confirm the supplier can support required documentation and process controls for the target category.
For example, in health and wellness products, FDA regulations may require tighter controls around materials, cleanliness, and recordkeeping. Even outside regulated categories, buyers should verify labeling workflows and test-report management to support customs and retailer requirements.
A realistic budget discussion also matters. Factory audit cost and factory audit pricing vary by scope, factory location, and technical depth, but the expense is usually small compared with rework, expedited freight, or a failed launch. For many importers, working with a China factory audits and evaluation service provider creates consistency across sites and gives management a clearer basis for go or no-go decisions.
Used this way, audits are not a one-time checkbox. They are a repeatable risk-control step that supports profitable, reliable China sourcing.
After identifying sourcing risk, brand exposure, and compliance pressure, the practical question becomes which audit type to run and when. In a successful China factory audit plan, each audit category checks a different failure point, so buyers should match scope to product risk, order value, and market requirements.
A Quality Management System audit examines how a factory controls quality across incoming materials, production, inspection, corrective action, and records. For importers, this is often the core factory audit because it predicts consistency before mass production starts. A strong review looks beyond certificates and tests whether procedures are used on the shop floor, especially for high-defect categories.
Focus on three practical checks: process control at key stages, traceability from raw material to finished goods, and nonconformance handling. If your team compares factory audit reviews across suppliers, this is usually where capability gaps become clear. This audit type also supports clearer factory audit sourcing decisions and helps justify factory audit cost by reducing rework, chargebacks, and delayed launches.
Social compliance audits verify labor practices, working hours, wage payment, age verification, grievance channels, and dormitory or canteen conditions where relevant. For US brands, this is critical because retailer requirements and customer expectations can trigger immediate reputational fallout if labor issues appear in your supply chain.
In execution, evaluate both documents and worker interviews, since policy alone does not confirm real practice. Align your checklist with your customer code of conduct and any category-specific obligations, especially when products may face stricter scrutiny in regulated sectors. When discussing factory audit pricing with a factory audit service, confirm whether interview sampling, multilingual auditors, and corrective action follow-up are included, since these affect depth and usefulness.
Environmental and safety audits assess waste handling, chemical storage, emissions controls, machine guarding, fire systems, and emergency readiness. For products sold in the US, weak EHS management can disrupt shipments, increase liability, and create compliance concerns, including risk around regulated materials. If your product touches health and wellness claims, maintain documentation discipline so production practices do not conflict with FDA-facing expectations.
On-site verification should include permit status, hazardous material segregation, PPE use, and evacuation route visibility. Ask the audit team to flag immediate hazards separately from systemic issues so remediation can be prioritized. Many importers use a phased approach: initial China factory audit for baseline risk, then targeted re-audits tied to corrective action deadlines and future purchase orders.
Across all three audit types, define pass criteria before booking the visit and require a corrective action plan with owners and due dates. That keeps your factory audit service outcome tied to purchasing decisions, not just a report file.
After reviewing quality systems, social compliance, and environmental safety, the practical question is execution. A strong China factory audit succeeds when preparation, fieldwork, and interviews are planned as one process tied to your sourcing goals, risk tolerance, and US market requirements.
Preparation determines whether your factory audit produces decisions or just observations. Before travel or booking a factory audit service, define the audit scope by product line, process stage, and critical risks such as labeling, traceability, and testing controls for US-bound goods. Build a checklist aligned to your purchase specifications, quality standards, and any applicable FDA-related expectations for health and wellness categories.
Collect and review core records in advance: business license, organization chart, production flow, quality manuals, calibration logs, corrective action history, subcontractor list, and recent inspection reports. Ask for digital copies before the visit so your team can flag gaps early. This also improves factory audit pricing discussions because a clear scope avoids unnecessary audit hours. For many buyers, factory audit cost varies mainly by audit depth, factory size, and whether specialist reviewers are required.
On-site work should validate documents against reality on the production floor. Start with an opening meeting that confirms agenda, access permissions, and photo policy. Then follow material flow from incoming inspection through packing and shipment release. Observe line setup, in-process controls, defect handling, and segregation of nonconforming goods.
Use short, structured checks to keep findings actionable:
Verify equipment status, calibration tags, and maintenance records.
Confirm worker instructions match the latest approved specification.
Review sampling plans, rework controls, and final release criteria.
Inspect warehouse practices, lot traceability, and packaging accuracy.
Test document control by tracing one order from PO to shipment file.
Capture objective evidence, including document IDs, timestamps, and photos where permitted. This improves internal factory audit reviews and helps sourcing teams compare factories consistently during factory audit sourcing decisions.
Interviews reveal whether procedures are understood and applied under normal production pressure. Speak with management about capacity planning, quality escalation, supplier controls, and corrective action ownership. Ask workers to describe their tasks, defect criteria, and stop-line authority in their own words. If answers differ from written procedures, treat that as a training or control risk.
Use separate interview tracks for supervisors and operators to reduce bias. Keep questions specific, neutral, and tied to observed processes. Close with a findings meeting that prioritizes issues by business impact, owner, and due date. For US importers, this step is essential because unresolved process gaps can lead to delays, chargebacks, or compliance exposure. If internal resources are limited, partnering with a China factory audits and evaluation service provider can improve consistency while keeping your audit program focused on measurable sourcing outcomes.
After preparing documents, running on-site checks, and interviewing managers and workers, your audit process needs one more decision: who should run and manage audits over time. The right operating model affects speed, consistency, and risk control, especially when your US team is buying from multiple China factories.
For many importers, the core choice is between building an internal audit function or hiring a China factory audit partner. In-house teams give you tight control over audit criteria, reporting format, and follow-up cadence. This can work well if you have recurring volume, trained staff in China, and clear category-specific standards.
A third-party factory audit service is often better when you need local coverage quickly, independent findings, or multilingual auditors across regions. It is also useful when procurement, quality, and compliance teams need objective reports before approving a new supplier. In practice, many US buyers use a hybrid model: third-party audits for supplier onboarding and periodic independent checks, with internal teams handling corrective action tracking.
When comparing options, focus on operational fit instead of brand size alone:
Scope clarity: Confirm whether the service covers quality systems, social compliance, process capability, and documentation review.
Reporting depth: Ask for sample reports with photos, nonconformance grading, and corrective action deadlines.
Turnaround and escalation: Define how fast urgent findings are reported and who receives alerts.
Cost structure: Review factory audit pricing by audit type, factory location, and auditor-day assumptions.
Integration: Check how findings feed into sourcing approval and supplier scorecards.
For budgeting, factory audit cost usually varies by audit scope and travel complexity, not only by provider name. Request line-item quotes in USD and confirm what is included, such as re-audit support, translation, and post-audit consulting.
Strong execution also depends on software. Whether you run in-house or outsourced audits, digital tools improve consistency and make factory audit reviews easier to compare across sites.
Look for software that supports mobile checklists, photo evidence, CAPA tracking, and role-based permissions. A practical setup links audit findings to supplier status so sourcing teams can block purchase orders until critical issues are closed. If you manage China factory audit programs from the US, cloud access and clear dashboard views are essential for weekly decision-making.
Common software categories include:
Audit workflow platforms for checklist execution, scoring, and approvals.
Quality management systems (QMS) for nonconformance, CAPA, and trend tracking.
Supplier management tools that combine qualification, audit history, and performance.
Before selecting a tool, run a pilot with one category and two or three suppliers. Measure reporting time, closure speed, and usability for both China-based auditors and US stakeholders. This keeps factory audit sourcing decisions practical and aligned with your broader supplier risk strategy.
If you need a starting point, working with a China factory audits and evaluation service provider that already uses structured digital workflows can reduce setup time and improve rollout discipline.
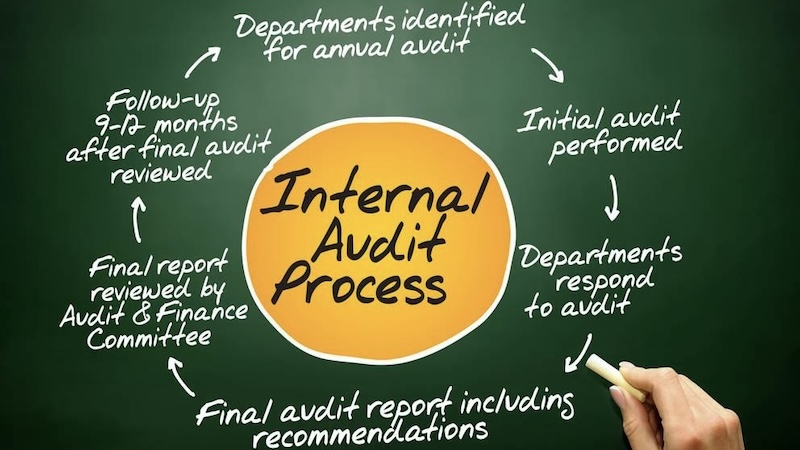
After deciding between internal teams, external partners, and software support, most US importers face a practical question: what will a China factory audit actually cost, and how should that budget be managed? Getting factory audit pricing right is not about choosing the cheapest quote. It is about paying for the level of verification that protects product quality, delivery timelines, and brand risk in the US market.
A realistic factory audit cost depends on scope, depth, and execution details rather than one fixed market rate. For a China factory audit, these variables usually drive price most:
Audit type and objective: A basic capability check is usually less expensive than a full social compliance, quality system, or technical process audit. If your products touch regulated categories in the US, such as health and wellness items with potential FDA scrutiny, the audit scope often expands and pricing follows.
Factory complexity: Multi-line production, subcontracting layers, and specialized equipment increase review time. More processes to verify means more auditor hours.
Location and logistics: A factory in a major industrial hub is generally more efficient to audit than one in a remote area requiring extra travel time and coordination.
Reporting requirements: Detailed corrective action plans, bilingual reports, photo evidence, and follow-up calls all add professional time.
Timeline urgency: Rush scheduling and compressed reporting windows typically increase factory audit pricing.
When comparing proposals, ask exactly what is included in the factory audit service: prep review, on-site hours, worker and management interviews, document checks, and post-audit support. This avoids confusion later and improves factory audit reviews across your sourcing team.
For US buyers managing factory audit sourcing across multiple SKUs, budgeting should be tied to risk and business impact, not only procurement pressure. A practical approach is to build an annual audit plan by supplier tier:
High-risk or high-value suppliers: Allocate budget for deeper initial audits and at least one follow-up verification.
Medium-risk suppliers: Plan standard audits with targeted checks on known weak points.
Low-risk repeat suppliers: Use lighter surveillance audits unless performance trends worsen.
Set your budget to cover both the first audit and corrective action verification. Many teams underfund follow-up work, which weakens audit value. It is also useful to reserve contingency funds when new factories are added quickly during peak purchasing cycles.
From a process standpoint, align payment approvals to your normal workflow, so audit scheduling is not delayed by finance bottlenecks. If you work with a China factory audits and evaluation service provider, confirm service levels in writing and map deliverables to sourcing milestones before purchase orders are finalized. Done well, factory audit pricing becomes a control tool that supports dependable launches, fewer quality surprises, and stronger supply continuity.
With cost drivers and budgeting now clear, the practical focus is decision quality. A successful China factory audit comes from matching scope, timing, and follow-up discipline to your product risk and US market requirements.
Define audit scope before scheduling. Tie each factory audit to product category risk, process complexity, and required compliance checks. For US-bound health and wellness products, include checkpoints relevant to FDA regulations so findings are decision-ready.
Separate price from value in vendor comparisons. Factory audit pricing should be evaluated against report depth, auditor capability, and evidence quality, not the lowest quote. A low factory audit cost that misses critical issues increases downstream risk.
Standardize pass, conditional pass, and fail thresholds. Use consistent decision rules across sites so factory audit reviews are comparable over time. This avoids subjective approvals and supports clearer sourcing decisions.
Build corrective action tracking into the plan. A factory audit service delivers full value only when non-conformities are assigned owners, deadlines, and verification steps. Repeat checks should confirm closure, not just restate earlier findings.
Integrate audits into factory audit sourcing workflows. Treat each China factory audit as part of supplier qualification and ongoing performance management, not a one-time event. This keeps audit outcomes aligned with quality, lead-time, and cost expectations.
In short, reliable outcomes come from disciplined scope definition, consistent evaluation criteria, and structured follow-through. That combination improves audit confidence while keeping factory audit cost and operational risk in balance.
Q1: What is the average cost of a factory audit in China?
The cost of a factory audit in China typically ranges from $300 to $1,500 per day, depending on the audit scope, factory location, and auditor expertise. Basic supplier verification audits are usually less expensive, while comprehensive social compliance or quality system audits cost more. Third-party audit firms often charge $500-$800 for a standard one-day audit. Additional costs may include travel to remote locations, translation services, and follow-up reports. While this seems like an upfront expense, it's significantly cheaper than dealing with defective products, production delays, or compliance violations that could cost tens of thousands of dollars in lost inventory, chargebacks, or legal penalties.
Q2: How long does a typical factory audit take in China?
A standard factory audit in China usually takes 6 to 8 hours for a single facility, typically completed in one full day. Simple supplier verification audits may take 3-4 hours, while comprehensive quality system audits or social compliance audits can extend to 1-2 days depending on factory size and complexity. The timeline includes document review, production floor inspection, worker interviews, and management meetings. Larger facilities with multiple production lines or buildings may require additional time. It's recommended to schedule audits during normal production hours to observe actual operating conditions. Follow-up audits to verify corrective actions are usually shorter, taking 2-4 hours.
Q3: When is the best time to conduct a factory audit in China?
The best time to conduct a factory audit is before placing your first significant order or committing to a long-term supplier relationship. Ideally, schedule the audit after initial samples meet your standards but before sending a deposit for mass production. This timing allows you to verify the factory's actual capabilities while still having negotiating leverage. Avoid auditing during Chinese New Year (late January to February) when factories operate with skeleton crews, or during peak production seasons when your audit may be rushed. It's also wise to conduct unannounced or short-notice audits to see genuine working conditions rather than staged presentations.
Q4: Can I conduct a factory audit in China myself or should I hire a third party?
Both options have merits depending on your experience and resources. Conducting your own audit gives you direct insight and builds supplier relationships, and it's cost-effective if you have staff in China with technical expertise and language skills. However, hiring a third-party auditor offers objectivity, professional expertise, standardized reporting, and local market knowledge that most importers lack. Third-party auditors also eliminate language barriers and can assess compliance issues more thoroughly. For first-time importers or high-risk products, professional auditors are recommended. Experienced importers often combine both approaches: using third-party audits for initial qualification and periodic self-audits for ongoing relationship management.
A successful China factory audit is not about finding a perfect supplier. It is about reducing preventable risk before you place larger POs, protecting your cash flow, and confirming that the factory can meet your quality and compliance expectations for the US market. If your product category touches health or wellness positioning, include checks tied to FDA regulations in your audit checklist so claims, labels, and controls stay aligned.
At this stage, treat your factory audit as a go or no-go gate. Define what must be true before approval, what can be corrected with a CAP (corrective action plan), and what automatically disqualifies a factory. This keeps decisions objective and prevents avoidable delays later in production.
Use this short action plan to close the process:
Lock your audit scope in writing. Include production capability, quality systems, social compliance, and product-specific controls.
Request transparent factory audit pricing before booking. Confirm what is included, what triggers extra fees, and expected deliverables.
Compare factory audit cost against order value and risk exposure. A lower upfront fee is not better if findings are shallow.
Review recent factory audit reviews for consistency in reporting quality, speed, and issue follow-up.
Schedule the audit and define acceptance criteria before deposit payment. Use payment methods your US finance team can track, such as Credit Cards, PayPal, or Apple Pay where applicable.
If you are still evaluating support options, a structured factory audit service can help standardize checklists, reporting, and corrective action tracking, especially during factory audit sourcing across multiple candidates. The key is to keep ownership of the decision criteria on your side.
Your concrete next step: choose one target factory, finalize your checklist today, and book the audit date within the next 5 business days. A scheduled audit with written pass/fail criteria turns this project from uncertain research into controlled progress.